You are here : Home City of energies > HOME BIAM > Search > EBMP research topics and projects
Research axes
1. From nutrient sensing to nutrient homeostasis and biomass growth
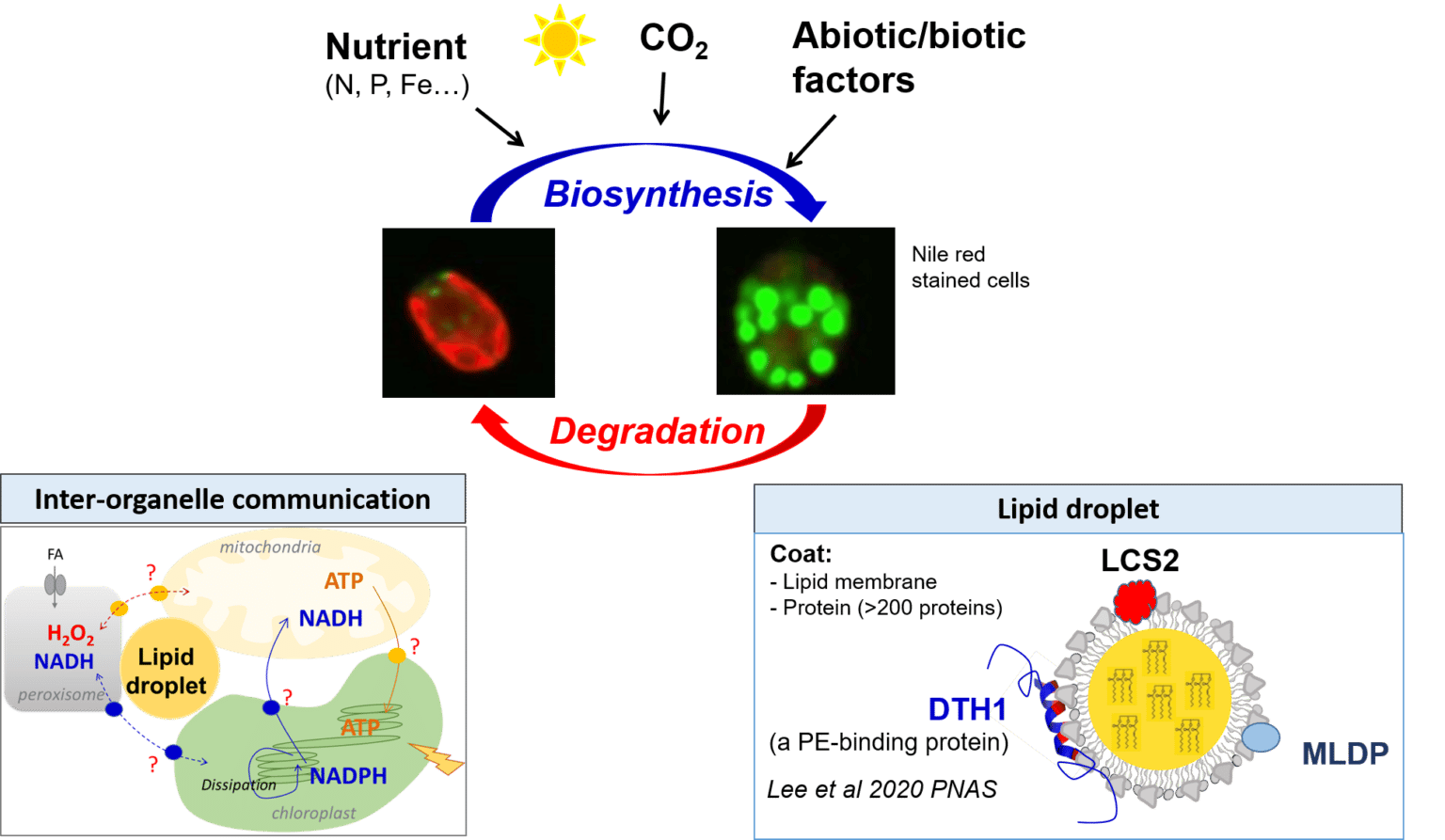
Photosynthetic growth relies on a balanced supply of solar energy, CO2 and nutrients (nitrogen, phosphate, iron…). Faced with a fluctuating environment (nutrient availability, temperature increase, water shortage, fluctuating CO2 levels…), cells must constantly adjust their metabolism to ensure their survival. In order to increase biomass productivity and make plants more resilient to a changing environment, it is necessary to understand the mechanisms underlying nutrient recovery/sensing/homeostasis and their importance in CO2 capture/utilization. Our main objectives are to dissect the interaction between carbon, iron, nitrogen and phosphate metabolic pathways in plants and algae (e.g., through the study of photorespiration and soil exploration by roots). To understand phosphate and iron responses and their impact on lipid homeostasis and triacylglycerol (TAG) storage, we will take particular advantage of state-of-the-art cell biology tools to dissect in vivo signaling and homeostasis of these nutrients.
2. What is photosynthesis?
Microalgae efficiently convert solar energy into biomass through photosynthesis, using water and carbon dioxide as their main resources. These microorganisms synthesize molecules of interest that can be used for the elaboration of biofuels (biohydrogen, biodiesel, biokerosene…) or biosourced compounds for chemistry.
The research developed in the laboratory aims to understand the biological mechanisms of conversion and storage of solar energy into energy-rich compounds (lipids, hydrocarbons, starch, hydrogen …) and to identify the locks for biotechnological applications.
2.1 Photosynthesis and bio-production of hydrogen

Photosynthesis is a highly regulated biological mechanism that converts solar energy and atmospheric CO2 into biomass. Our research aims at understanding the regulation of photosynthesis in order to domesticate this process for an optimized production of biomass or energy-rich compounds (hydrogen, starch, lipids…). The research is mainly conducted on the model microalga Chlamydomonas reinhardtii.
2.2 Regulation of photosynthesis
Forward genetic approaches based on high-throughput screening of mutants are developed to identify mutants affected in novel mechanisms of photosynthesis regulation. Different screens, based on the analysis of chlorophyll fluorescence or on the analysis of carbon storage capacity, have been set up in the laboratory. The phenotypic analysis of mutants by multidisciplinary approaches allows to understand the physiological function of genes of interest.
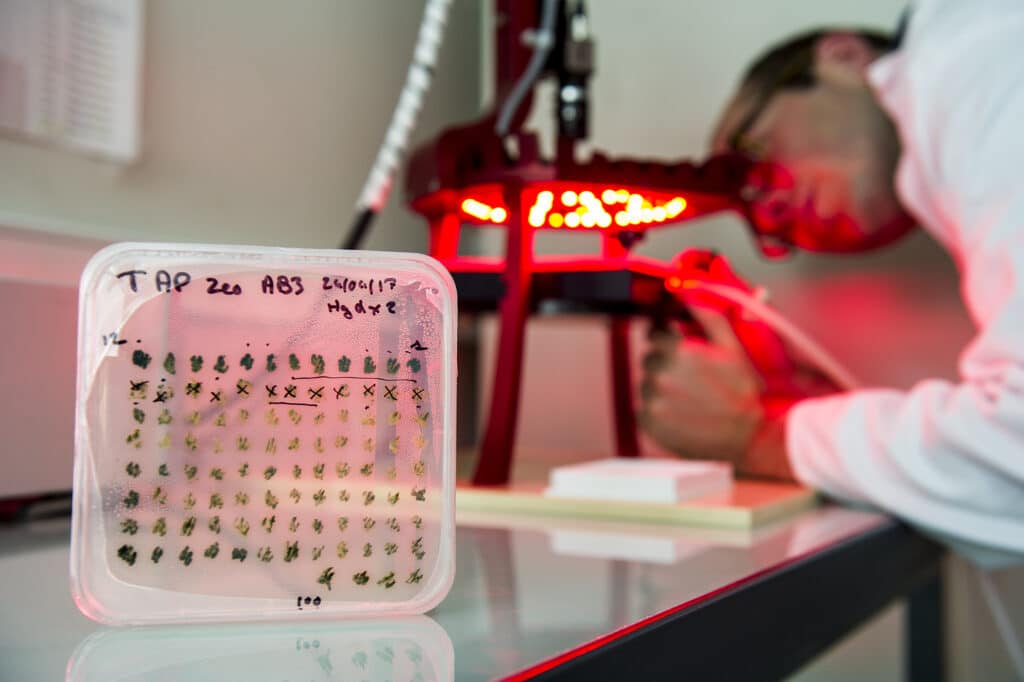
2.3 Photoproduction of hydrogen
Some microalgae (such as Chlamydomonas reinhardtii) have the ability to produce hydrogen through a hydrogenase interacting with the photosynthetic electron transport chain. In nature, hydrogen photo-production is a transient phenomenon used to evacuate the excess reducing power generated. The research aims at understanding the electron transfer pathways involved in hydrogen photoproduction, their regulation and limitations, in order to develop biotechnological applications.
3. Lipid metabolism and triacylglycerol storage
Microalgae have a strong capacity to synthesize fatty acids and to accumulate them in the form of oil reserves (up to 50% of their dry weight). Fatty acids (or derivatives) extracted from algal biomass have important potential applications in biofuels (biodiesel, biopetroleum), human nutrition (polyunsaturated fatty acids called omega 3) and green chemistry (synthons for biopolymers, lubricants, etc.). However, major biological barriers remain to be removed to ensure the profitability of the sector. For example, the accumulation of oil in microalgae only occurs under stressful conditions, such as nutrient deficiency, which limits cell growth and therefore biomass and oil productivity. Moreover, biofuel and human nutrition applications require an improvement of the “quality” of the microalgae oil, i.e. an optimization of the fatty acid composition, adapted to each case.
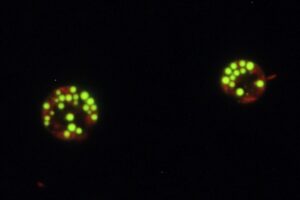
The aim of this research is to understand the synthesis pathways of reserve lipids (triacylglycerols) and their regulation in relation to environmental conditions (light, nitrogen deficiency, etc.) and energy metabolism (photosynthesis). They are conducted mainly in the model microalgae Chlamydomonas reinhardtii. The work is based on three complementary approaches:
i) a forward genetic approach aiming at isolating mutants affected in their capacity to accumulate or destock oil under various culture conditions (in particular under optimal growth conditions), as well as mutants whose fatty acid profile is modified. The mutants are screened by high-throughput methods (flow cytometer, GC-MS with automated sample preparation). After identification of genes of interest and fine characterization of the isolated mutants by lipidomic analysis (LC-MS/MS), innovative strategies to improve oil content and quality are proposed.
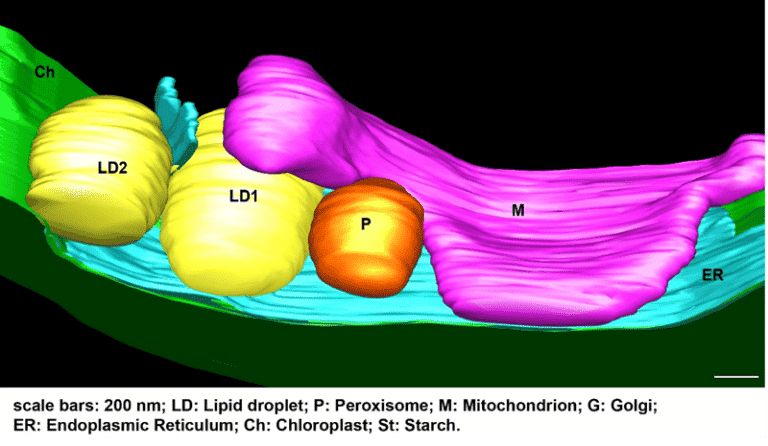
ii) Lipid droplet biology and dynamics. This approach aims to study the proteins that are associated to lipid droplet and discover their biochemical and physiological functions.
iii) a third approach is based on a reverse genetic method. Knock-out mutants for desirable candidates will either be ordered from the CLIP collection, or generated using Crispr-cas9 technology.


4. Biosynthesis and function of hydrocarbons (alkanes, alkenes)
Like many organisms, microalgae and cyanobacteria are able to synthesize hydrocarbons from the fatty acids constituting their lipids (membrane or reserve). Linear alkanes and alkenes are important components of fossil fuels. Their synthesis and excretion in the culture medium would allow to avoid the costly steps of biomass harvesting, oil extraction and conversion into biodiesel. The research carried out in the laboratory aims at i) elucidating the biological function of hydrocarbons (alkanes, alkenes) naturally produced by microalgae and cyanobacteria ii) identifying the enzymes allowing the biosynthesis of hydrocarbons iii) studying the properties, the mechanism and the structure-function relationships of these enzymes and iv) exploiting the whole of this knowledge to explore biotechnological applications aiming at producing biosourced hydrocarbons. In particular, our work has identified the first microalgal enzyme for hydrocarbon synthesis (fatty acid photodecarboxylase, FAP).
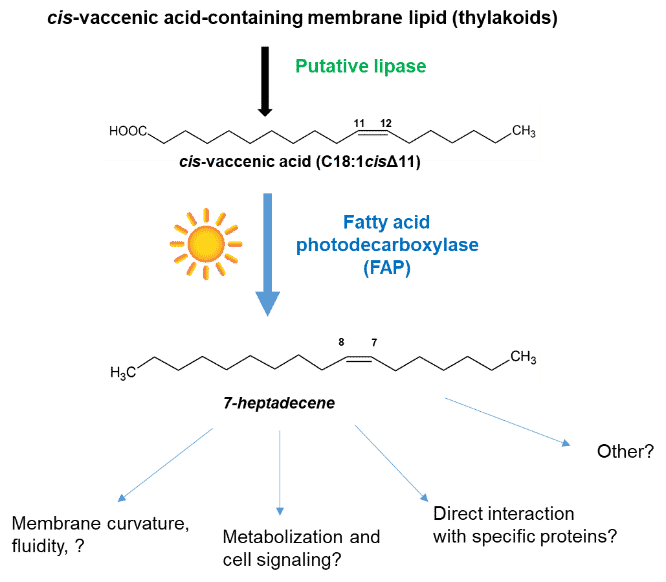
Our research on the enzymatic conversion of fatty acids also extends to enzymes allowing the functionalization of fatty acids. This work, also carried out with a view to applications in green chemistry, aims to identify new enzymes in microalgae or plants allowing the introduction of functional groups (hydroxy, epoxy, etc.) at particular positions of the carbon chain of fatty acids.
Team manager
Key words
Lipid; Lipidomics; Fatty acids; Lipid droplet; Hydrocarbons
alkanes; Bio-hydrogen; Photosynthesis; Photoenzyme
Energy trafficking; Carbon metabolism; Genetic engineering; Synthetic biology; Chlamydomonas reinhardtii; Nannochloropsis sp